- Home
- Services
- About
- News
- Contact
- Near lock mac download
- Layers of fear 3 children
- Snapgene confocal microscopy
- Deepest sword in xvideos lewd ninja
- Fun templates for google docs
- Best netflix movies
- Youtube linda rontadt hasten down the wind
- Set timer 12 minutes
- Diffrent typ o dimonad structers
- Gobot pokemon
- Sara leonard sbart
- Galaxy on fire 2 all blueprints
- Mk 19 grenade launcher
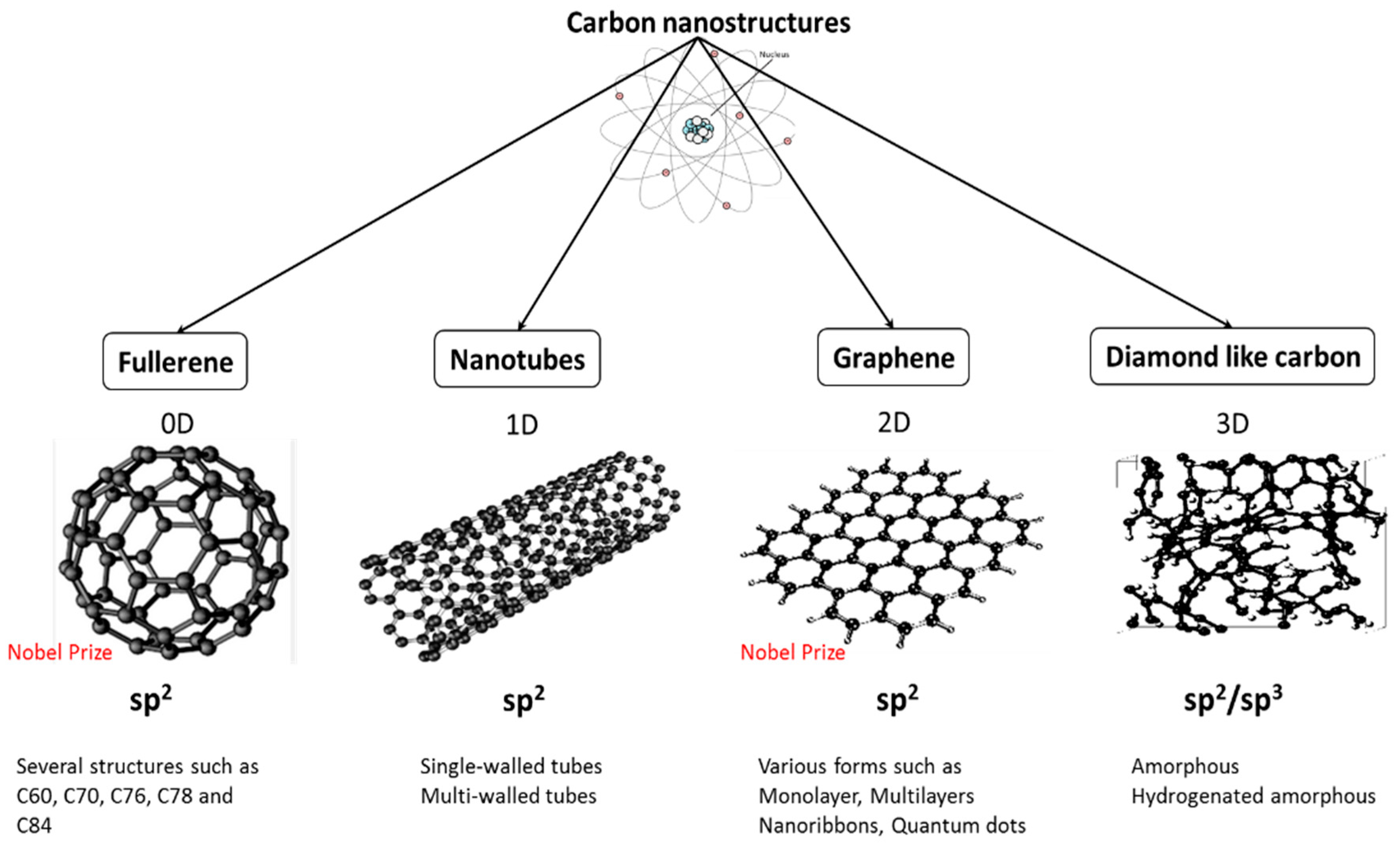
The layers, of course, extend over huge numbers of atoms - not just the few shown above. The distance between the layers is about 2.5 times the distance between the atoms within each layer. In that case, it is important to give some idea of the distances involved. Notice that you can't really draw the side view of the layers to the same scale as the atoms in the layer without one or other part of the diagram being either very spread out or very squashed. The diagram below shows the arrangement of the atoms in each layer, and the way the layers are spaced. Graphite has a layer structure which is quite difficult to draw convincingly in three dimensions. There are no possible attractions which could occur between solvent molecules and carbon atoms which could outweigh the attractions between the covalently bound carbon atoms. Is insoluble in water and organic solvents.
Diffrent typ o dimonad structers free#
All the electrons are held tightly between the atoms, and aren't free to move. This is again due to the need to break very strong covalent bonds operating in 3-dimensions.ĭoesn't conduct electricity. Very strong carbon-carbon covalent bonds have to be broken throughout the structure before sublimation occurs. Sublimes at ordinary pressures at almost 4000☌. Practise until you can do a reasonable free-hand sketch in about 30 seconds. In the case of diamond, each carbon is bonded to 4 other carbons rather than hydrogens, but that makes no essential difference.ĭon't try to be too clever by trying to draw too much of the structure! Learn to draw the diagram given above. If you are interested in a more modern view, you could read the page on bonding in methane and ethane in the organic section of this site. That simple view is perfectly adequate to explain the bonding in diamond. Note: We quoted the electronic structure of carbon as 2,4. It is not a molecule, because the number of atoms joined up in a real diamond is completely variable - depending on the size of the crystal. This is a giant covalent structure - it continues on and on in three dimensions. We are only showing a small bit of the whole structure.

In the diagram some carbon atoms only seem to be forming two bonds (or even one bond), but that's not really the case. In diamond, each carbon shares electrons with four other carbon atoms - forming four single bonds. This page describes the structures of giant covalent substances like diamond, graphite and silicon dioxide (silicon(IV) oxide), and relates those structures to the physical properties of the substances.Ĭarbon has an electronic arrangement of 2,4.
- Home
- Services
- About
- News
- Contact
- Near lock mac download
- Layers of fear 3 children
- Snapgene confocal microscopy
- Deepest sword in xvideos lewd ninja
- Fun templates for google docs
- Best netflix movies
- Youtube linda rontadt hasten down the wind
- Set timer 12 minutes
- Diffrent typ o dimonad structers
- Gobot pokemon
- Sara leonard sbart
- Galaxy on fire 2 all blueprints
- Mk 19 grenade launcher
