- Home
- Services
- About
- News
- Contact
- Near lock mac download
- Layers of fear 3 children
- Snapgene confocal microscopy
- Deepest sword in xvideos lewd ninja
- Fun templates for google docs
- Best netflix movies
- Youtube linda rontadt hasten down the wind
- Set timer 12 minutes
- Diffrent typ o dimonad structers
- Gobot pokemon
- Sara leonard sbart
- Galaxy on fire 2 all blueprints
- Mk 19 grenade launcher
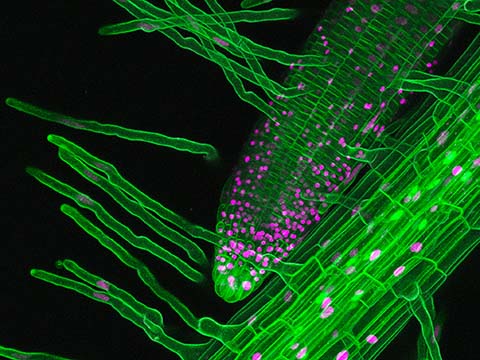
natans and no SN) treatment and V–S+ (no V. natans and SN) treatment compared with the V + S- (V. Lefse analysis showed that Saccharimonadales, Micrococcales, Sphingobacteriales, Bacteroidales, Obscuribacterales, Flavobacteriales, Pseudomonadaceae and Myxococcales, which are considered to be SN-resistant bacteria, increased significantly in the V + S+ (V. natans led to higher relative abundances of Saccharimonadales and Rhizoniales. natans significantly improved the SN removal efficiency likely by increasing DO, ORP and bacterial diversity in the water column. natans on sulfonamide removal in an additional 13-day exposure experiment with focus on the dynamics of dissolved oxygen (DO), the oxidation-reduction potential (ORP) and microbial communities in the water column, as well as in the periphyton on V. natans, the plant was overall resistant to the SN doses that we used. Although high SN concentrations had toxic effects on the growth of V.

natans was found under SN stress, but SN had only a weak effect on root length. After 20 days’ exposure, a reduction in the relative growth rate (RGR) and leaf length of V. Meanwhile, the levels of reactive oxygen species (ROS, including O2− and H2O2) and peroxidase (POD) increased with increasing SN concentration and duration of exposure. The toxic effect and oxidative stress caused by SN resulted in a reduction of total chlorophyll (chl.a and b) and autofluorescence of chloroplast. We studied both the ecotoxicological effects of sulfonamide (SN) on and its removal by Vallisneria natans (Lour.) Hara, an important submerged macrophyte in Chinese lakes and rivers. In aquatic environments, macrophytes may not only be affected by various pollutants, they may also help to reduce the concentrations in the surrounding environment. The extensive application of sulfonamides (SAs) raises concern regarding its negative environmental effects. (C) original magnification ×50,000 bar = 100 nm (D) original magnification ×20,000 bar = 250 nm).
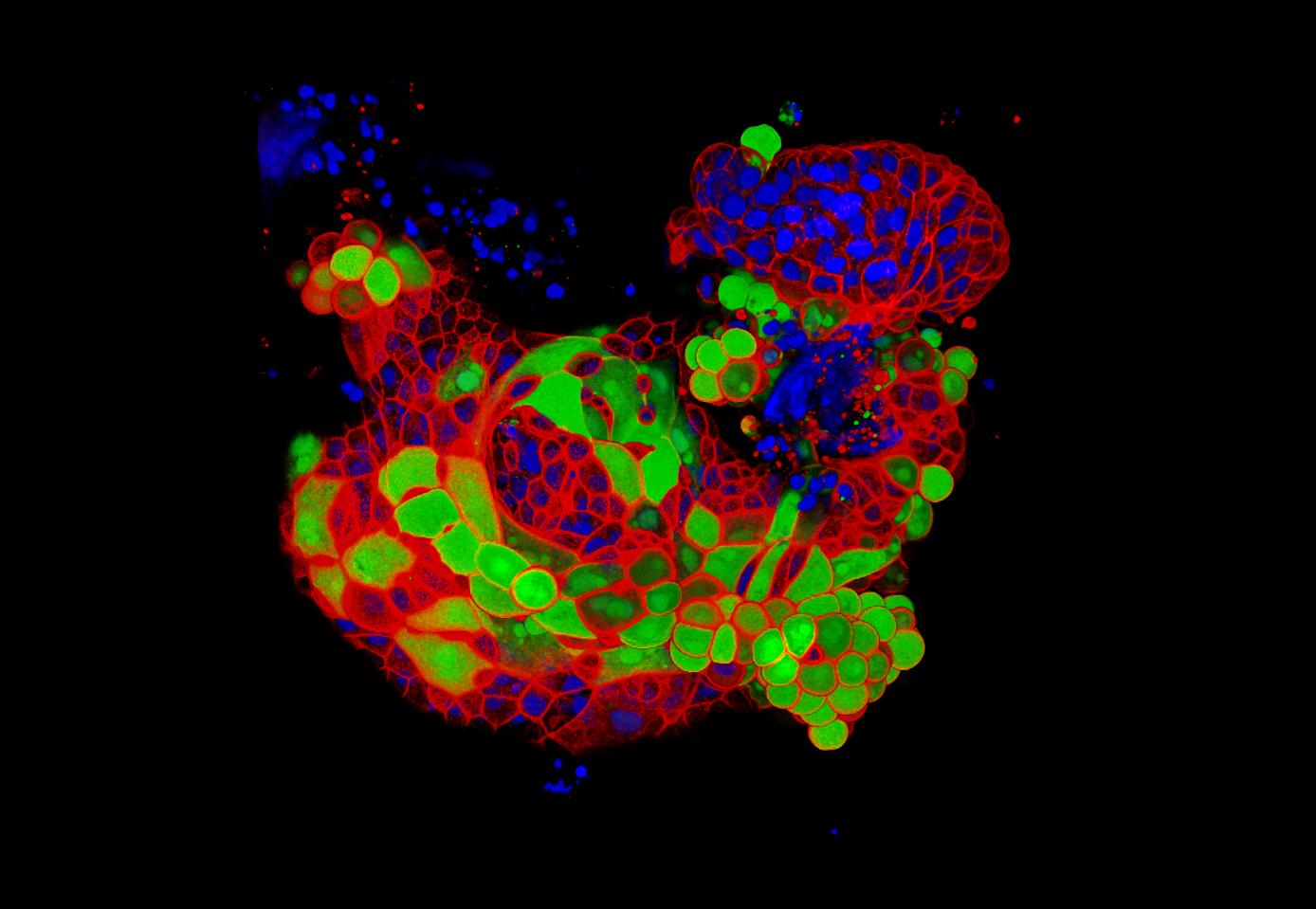
(C, D) LR Gold-embedded tissue: Note very dense specific immunolabeling and virtually complete absence of background. (Original magnification ×50,000 bar = 100 nm). (B) Araldite-embedded tissue (no postfixation): Note much lower immunolabeling density (at 1:100 antibody dilution) compared with the LR Gold samples (at 1:1000 antibody dilution) and rather high nonspecific background over the cytoplasm. Note very low level of specific labeling (over neuropeptide-containing granule).

(A): Araldite-embedded tissue additionally postfixed in 1% osmium tetroxide for 30 min at 4☌. The 10 nm protein A-gold probe was used throughout. (Sections from araldite samples were etched for 1 hour in saturated aqueous sodium metaperiodate before immunolabeling). All sections were immunolabeled with the same (polyclonal rabbit antineuropeptide) antibody (araldite samples at a dilution of 1:100 and the LR Gold sample at 1:1000). Araldite samples were resin-infiltrated at room temperature and then heat-polymerized. The LR Gold sample was processed as described herein. In all cases, the tissue was fixed initially in a mixture of 1% formaldehyde and 2.5% glutaraldehyde for 4 h at 4☌. Images (digital TEM) of sections from (snail auricle) tissue prepared by three different procedures (from which the data in Table 2 was derived). Comparative Immunolabeling Densities with Different Preparative Procedures.
- Home
- Services
- About
- News
- Contact
- Near lock mac download
- Layers of fear 3 children
- Snapgene confocal microscopy
- Deepest sword in xvideos lewd ninja
- Fun templates for google docs
- Best netflix movies
- Youtube linda rontadt hasten down the wind
- Set timer 12 minutes
- Diffrent typ o dimonad structers
- Gobot pokemon
- Sara leonard sbart
- Galaxy on fire 2 all blueprints
- Mk 19 grenade launcher
